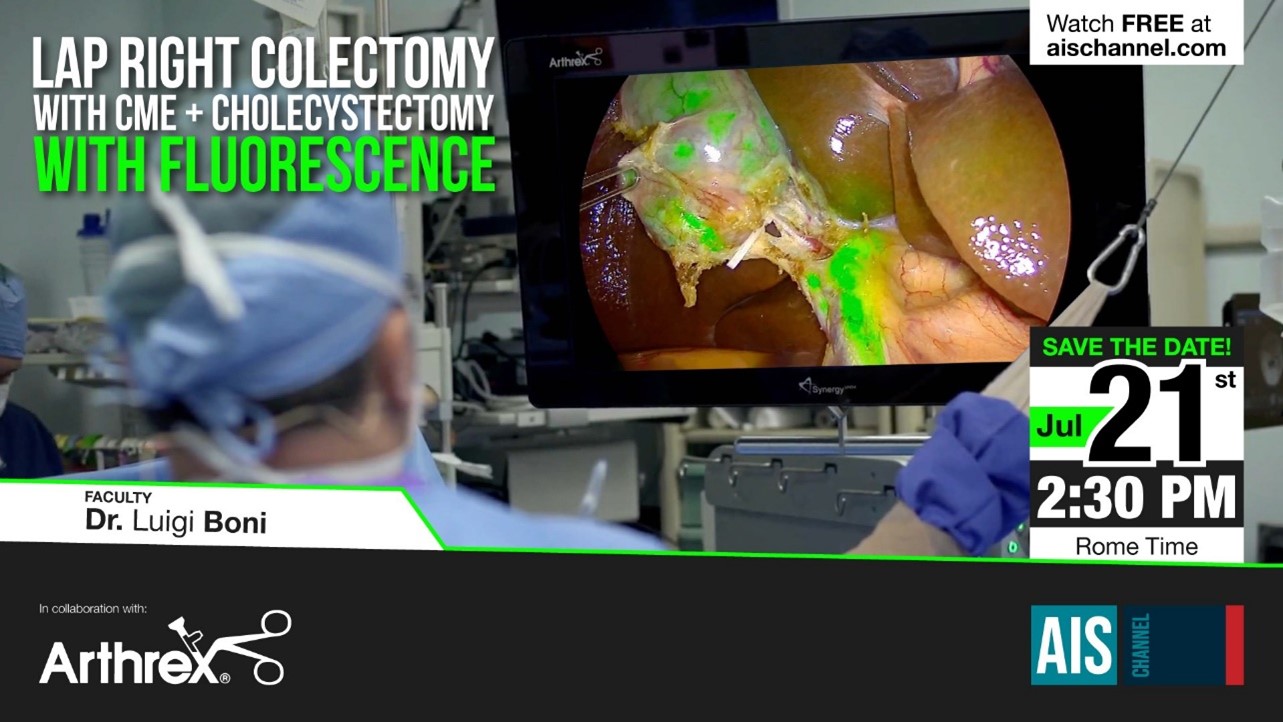
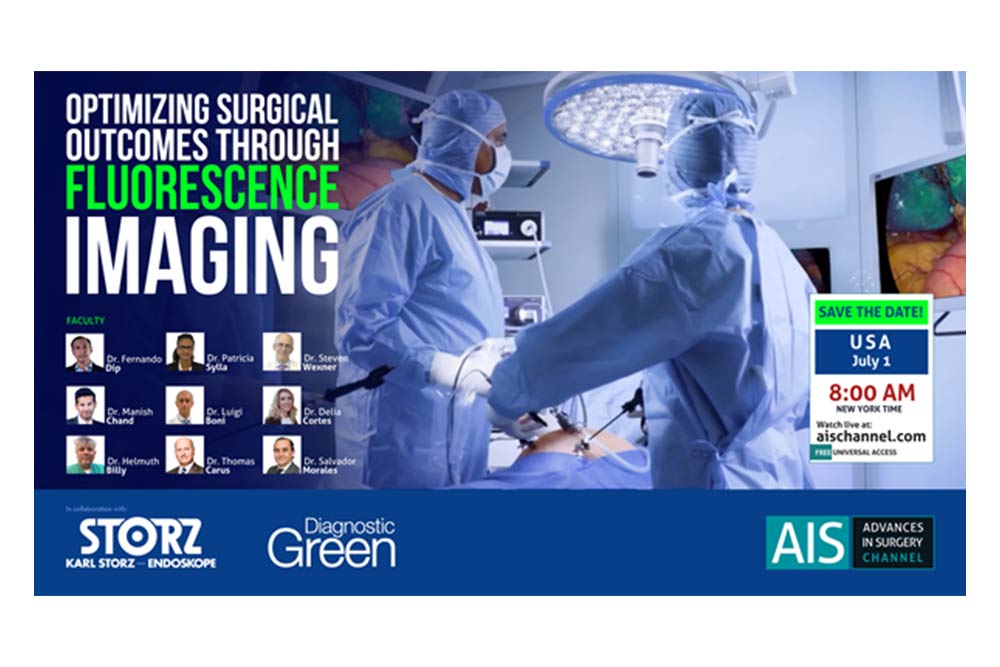
Diagnostic Green are delighted to support this free masterclass on The Role of ICG in Bariatric and Emergency General Surgery and its Role in Education and Training.
On 19th July at 7pm BST (8pm CET; 2pm US Eastern Standard Time) we will hear from Prof Boni from Italy, Prof Carus from Germany, Consultant surgeons Mr Awan and Bhatti from UK and Jo Cook from Kimal – Diagnostic Green’s rep in UK. A great and informative webinar promised – don’t miss out.
To register for this unique free event covering Emergency surgery use for ICG, click the following link.